PROFESSIONAL MEDICAL PRODUCTS
ASPIRATORE SUPER VEGA BATTERY PLUSSUPER VEGA BATTERY PLUS SUCTION UNITASPIRATEUR SUPER VEGA BATTERY PLUSSAUGER SUPER VEGA BATTERY PLUSASPIRADOR SUPER VEGA BATTERY PLUSASPIRADOR SUPER VEGA BATTERY PLUSSSAK AKUMULATOROWY SUPER VEGA BATTERY PLUSASPIRATOR SUPER VEGA BATTERY PLUSΣΥΣΚΕΥΗ ΑΝΑΡΡΟΦΗΣΗΣ SUPER VEGA BATTERY PLUS
28190
Gima S.p.A.Via Marconi, 1 – 20060 Gessate (MI) Italy[email protected] – [email protected]www.gimaitaly.comMade in Italy
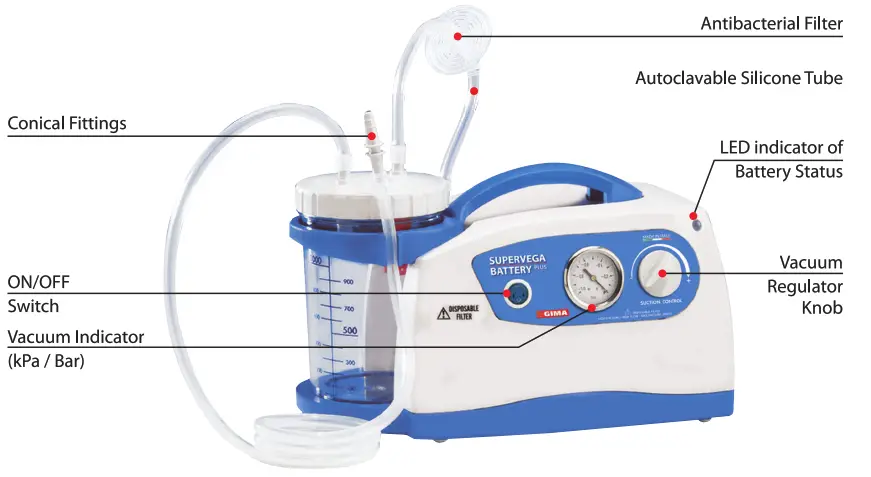
SUPER VEGA BATTERY PLUS SUCTION UNIT is a suction pump particularly suited for use in hospital wards, in patients with a tracheotomy, in small surgical applications, and in post-operative treatments at home. A device that can be used for nasal, oral, and tracheal aspirations in adults or for body liquids in children (for example mucus, phlegm, and blood). A device designed to offer ease-of- transport and almost continuous use thanks to the adoption of an electronic system to manage the power supply. Supplied with an audible alarm and visual tell-tale (luminous LED) to indicate the battery status. Featuring a body in plastic with high thermal and electrical insulation in compliance with recently introduced European safety regulations. Supplied complete with sterilizable polycarbonate jug with overflow valve. Features a suction regulator and vacuum gauge located on the front panel.
GENERAL WARNING
Read the instruction manual carefully before use. The device is for use by qualified personnel (surgeon / professional nurse/assistant) the use of the device at home is restricted to an adult in full possession of mental faculties and/or home carers the instrument must not be disassembled. For technical service always contact Gima S.p.A.?
IMPORTANT SAFETY RULES
- Check the condition of the unit before each use. The surface of the unit should carefully be inspected for visual damage. Check the mains cable and do not connect to power if damage is apparent;
- Before connecting the appliance always check that the electric data indicated on the data label and the type of plug used, correspond to those of the mains electricity to which it’s to be connected;
- Respect the safety regulations indicated for electrical appliances and particularly:– Use original components and accessories provided by the manufacturer to guarantee the highest efficiency and safety of the device;– The device can be used only with the bacteriological filter;– Never immerge the appliance into water;– Do not place or store the aspirator in places where it may fall or be pulled into the bathtub or washbasin.In the event it is accidentally dropped, do not attempt to remove the device from the water whilst the plug is still connected: disconnect the mains switch, remove the plug from the power supply and contact theGIMA technical service department. Do not attempt to make the device work before it has been thoroughly checked by qualified personnel and/or theGIMA technical service department.– Position the device on stable and flat surfaces in a way that the air inlets on the back aren’t obstructed;– To avoid incidents, do not place the aspirator on unstable surfaces, which may cause it to accidentally fall and lead to a malfunction and/or breakage. Should there be signs of damage to the plastic parts, which may expose inner parts of the energised device, do not connect the plug to the electrical socket. Do not attempt to make the device work before it has been thoroughly checked by qualified personnel and/or theGIMA technical service department.– Don’t use in the presence of inflammable substances such as anaesthetic, oxygen or nitrous oxide;– Don’t touch the device with wet hands and always prevent the appliance coming into contact with liquids;– Don’t leave the appliance connected to the power supply socket when not in use;– Don’t pull the power supply cable to disconnect the plug remove the plug from the mains socket correctly;– Store and use the device in places protected against the weather and far from any sources of heat. After each use, it is recommended to store the device in its own box away from dust and sunlight.– In general, it is inadvisable to use single or multiple adapters and/or extensions. Should their use be necessary, you must use ones that are in compliance with safety regulations, however, taking care not to exceed the maximum power supply tolerated, which is indicated on the adapters and extensions.– Prevent children from using the device without proper supervision;– Never leave the appliance near water, do not immerse it in any liquid. If the device has fallen into water, unplug it before you hold it. Do not use the appliance if the plug or AC / DC power supply is damaged or wet (send it immediately to an authorized service center or technical service).
- For repairs, exclusively contact technical service and request the use of original spare parts. Failure to comply with the above can jeopardize the safety of the device;
- Use only for the purpose intended. Don’t use for anything other than the use defined by the manufacturer.The manufacturer will not be responsible for damage due to improper use or connection to an electrical system not complying with current regulations.
- The medical device requires special precautions regarding electromagnetic compatibility and must be installed and used in accordance with the information provided with the accompanying documents: the SUPER VEGA BATTERY PLUS SUCTION UNIT device must be installed and used away from mobile and portable RF communication devices (mobile phones, transceivers, etc.) that may interfere with the said device.
- Instrument and accessory discharging must be done according to current regulations in the country of use.
- WARNING: Do not change this equipment without the permission of the manufacturer GIMA S.p.A. None of electric or mechanical parts has been designed to be repaired by customers or end-users. Don’t open the device, do not mishandle the electric/mechanical arts. Always contact technical assistance
- Using the device in environmental conditions different than those indicated in this manual may harm seriously the safety and the technical characteristics of the same.
- The medical device is in contact with the patient by means of a disposable probe (not supplied with the device). Suction tubes for insertion in the human body purchased separately from the machine should comply with ISO 10993-1 standards on material biocompatibility.
- The product and its parts are biocompatible in accordance with the requirements of regulation606011.
- Operation of the device is very simple and therefore no further explanations are required other than those indicated in the following user manual.
- The lead battery integrated in the device is not to be considered as an ordinary domestic aste. Such a component must be disposed of in a specific collection centre in order to be recycled.
- Use in Home-Care: Keep all accessories of the device out of reach of children under 36 months of age since they contain small parts that may be swallowed.
- Do not leave the device unattended in places accessible to children and/or persons not in full possession of mental faculties they may strangle themselves with the patient’s tube and/or the power cable.
The manufacturer cannot be held liable for accidental or indirect damages should the device be modified, repaired without authorization or should any of its components be damaged due to accident or misuse. Any minimal modification/repair on the device voids the warranty and does not guarantee compliance with the technical requirements provided by the MDD 93/42/EEC (and subsequent changes) and its normatives.
CONTRAINDICATIONS
– Before using the SUPER VEGA BATTERY PLUS SUCTION UNIT, consult the instructions for use: failure to read all the instructions in this manual can be harmful for the patient.– The device cannot be used to drain chest fluids;– The device must not be used for suction of explosive, corrosive or easily flammable liquids.– SUPER VEGA BATTERY PLUS SUCTION UNIT is not suitable for MRI. Do not introduce the device in MRI environments.
TECHNICAL CHARACTERISTICS
|
PROBLEM |
SOLUTION |
| No power | Insert the plug securely into the power outlet. |
| No picture | Ensure that the equipment is connected properly. |
| Ensure the correct input is selected. | |
| Buttons on the monitor don’t do anything | Disconnect the power plug and then reconnect it. (The unit may not be operating properly due to lightning, static, or other factors.) |
| Noise or other interference | Electronic equipment near the unit may be causing interference. Relocate the unit or move the equipment. |
The technical specifications may change without notice.
CLEANING OF THE DEVICE
Use a soft dry cloth with not – abrasive and not – solvent detergents. To clean the device external parts always use a cotton cloth dampened with detergent. Don’t use abrasive or solvent detergents. Before carrying out any cleaning and/or maintenance operation, disconnect the appliance from the power supply, unplug it or turn off the switch on the device.
Particular care should be taken to ensure that the internal parts of the equipment do not get in touch with liquids. Never clean the equipment underwater.During all clearing operations use protective gloves and apron (if need be, also wear a face mask and glasses) to avoid getting in contact with contaminating substances (after each utilization cycle of themachine).
ACCESSORIES SUPPLIES
- Complete aspiration jar 1000ml
- Conical fitting
- Tubes set 8 mm x 14 mm
- Hydrophobic and antibacterial filter
- AC/DC adapter
- Power supply cord for AC/DC adapter
- Cigarette ligther cable
Available under request with different versions with complete jar 2000ml.
The filter is produced with (PTFE) hydrophobic material to prevent fluids from entering the pneumatic circuit. It should be changed immediately if it becomes wet or if there is any sign of contamination or discoloration. If should also be changed if the unit is used with a patient whose risk of contamination is unknown. Don’t use the suction unit without the protection filter. If the suction unit is used in an emergency or in a patient where the risk of contamination is not known the filter must be changed after each use. The filter is not designed to be decontaminated, disassembled, and/or sterilized. If the patient’s pathology is known and/or no risk of indirect contamination exists, it is advisable to replace the filter after each work shift or at any rate on a monthly basis even if the device is not used.
WARNING: Suction tubes for insertion in the human body purchased separately from the machineshould comply with ISO 10993-1 standard on material biocompatibility.
Aspiration jar: the mechanical resistance of the component is guaranteed up to 30 cycles of cleaning and sterilization. Beyond this limit, the physical-chemical characteristics of the plastic material may show signs of decay. Therefore, we recommend that you change it.
Silicone tubes: the number of cycles of sterilization and/or cleaning is strictly linked to the employment of the said tube. Therefore, after each cleaning cycle, it is up to the final user to verify whether the tube is suitable for reuse. The component must be replaced if there are visible signs of decay of the material constituting the said component.
Conical fitting: the number of cycles of sterilization and the number of cleaning cycles is strictly linked to the employment of the said component. Therefore, after each cleaning cycle, it is up to the final user to verify whether the fitting is suitable for reuse. The component must be replaced if there are visible signs of decay in the material constituting the said component.
Service life of the device: more than 1000 hours of operation (or 3 years) in accordance with the standard conditions of testing and operation. Shelf life: maximum 5 years from the date of manufacture.
WARNING: The medical device is provided without a specific suction probe. If this device must beused with a specific suction probe, the end-user is responsible for making sure it complies with theEN 10079-1 regulation.
CLEANING OF ACCESSORIES
Before using the device, the manufacturer advises you to clean and/or sterilize the accessories. Washing and/or cleaning the autoclavable jar is to be carried out as follows:
- Wear protective gloves and apron (glasses and face mask if necessary) to avoid contact with contaminating substances.
- Disconnect the tank from the device and remove the said container from the support of the device.
- Separate all the parts of the cover (overflow device, washer).
- Disconnect all tubes from the jar and the protection filter.
- Empty and dispose of the contents of the suction vessel (also comply with regional regulations);
- Wash each part of the container from secretions under cold running water and then clean every single part in hot water (temperature not exceeding 60°C). Once again, carefully wash every single part using, if necessary, a non-abrasive brush to remove any deposits.
- Rinse with hot running water and dry all parts with a soft cloth (non-abrasive).
- Dispose of the aspiration catheter according to the provisions of local laws and regulations.
The jug and lid can be further disinfected using a common disinfectant, strictly following the instructions and dilution values provided by the manufacturer. At the end of cleaning operations, leave to air dry in a clean environment.
The silicone aspiration tubes and the conical fitting may be carefully washed in hot water (temperature must not exceed 60°C). After cleaning, leave the parts to dry in an open, clean environment. When cleaning is complete, reassemble the container for liquid aspirations according to the following procedure:
- Place the overflow valve into its seat in the cover (under VACUUM connector)
- Insert floating valve keeping the o-ring towards the opening of the cage
- Place the o-ring into its seat around the cover
- After completing assembling operations always make sure that the cover seals perfectly to avoid vacuum leakages or liquid exit
The jar and the cover can be autoclaved by placing the parts into the autoclave and running one sterilization stem cycle at 121°C (1 bar relative pressure – 15 min) making sure that the jar is positioned upsidedown. Mechanical resistance of the jar is guaranteed up to 30 cycles of sterilization and cleaning at the indicated conditions (EN ISO 10079-1). Beyond this limit, the physical-mechanical characteristics of the plastic may decrease and replacement of the part is therefore recommended.
After sterilization and cooling at an environmental temperature of the parts make sure that these are not damaged. The aspiration tubes can be sterilized on autoclave using a sterilization cycle at 121°C (1 bar relative pressure – 15 min). The conical connector can be sterilized on autoclave using a sterilization cycle at 121°C (1 bar relative pressure – 15 min).
DO NOT WASH, STERILIZE OR PUT IN AUTOCLAVE THE ANTIBACTERIAL FILTER
PERIODICAL MAINTENANCE CHECKS
The SUPER VEGA BATTERY PLUS SUCTION UNIT suction equipment does not need maintenance or lubrication. It is, however, necessary to inspect the unit before each use. With regard to training, given the information contained in the user manual and since it is easy to understand the said device, it doesn’t appear to be necessary. Unpack the instrument and always check the integrity of plastic parts and AC/DC switching adapter, feeding cable, they might have been damaged during previous use. Connect the cable to the electrical network and turn the switch on. Close the aspirator outlet with your finger and with the suction regulator in maximum vacuum position check that the vacuum indicators reach -80kPa (-0.80 bar) minimum (internal battery). Rotate the knob from right to left and check the aspiration regulating control. The vacuum indicator should go down -40kPa (-0.40 bar). Verify that loud noises are not present, these can indicate wrong functioning. The device is protected by a safety fuse (F 10A L 250V) situated in the cigarette lighter cable. When replacing, always check the type and value as indicated.Internally, the device is protected (see electrical specifications) by two fuses F1, F2 (T 15A L 125V) that cannot be reached from the outside. Therefore, contact the manufacturer to request the assistance of an authorized and qualified technician when they need to be replaced. If it’s replaced make sure that its replacement is always the same type and value, as indicated. The device is made up of a lead battery that cannot be accessed from the outside. In order to replace it, consult the technical staff authorized by the manufacturer.
USE ONLY THE RECOMMENDED BATTERIES FROM GIMA. THE USE OF OTHER BATTERIESIS NOT RECOMMENDED AND INVOLVES THE CANCELLATION OF THE WARRANTY
In the event that the service personnel has to replace the internal battery, pay special attention to the polarity of the same component. The + / – polarities are indicated directly on the battery.
|
Fault type |
Cause |
Solution |
| 1. Red light on | Battery run down | Hook up the power cord to the electricity mains, positioning the equipment power switch on 0. |
| 2. No light | Defective AC/DC adapter or technical internal problem | Contact the technical service. |
| 3. No aspiration | Jar Cap badly screwed down | Unscrewed the cap, then rescrew it correctly |
| 4. No aspiration | Lid seal not in its seat | Unscrew the cap and insert the seal properly in its seat |
| 5. The float doesn’t close | If the cap has been washed, ensure that the float is not partially detached | Insert the float into it’s place |
| 6. The float doesn’t close | The float it’s covered by dirty material | Unscrewed the cap, leave the and put in on au- toclave |
| 7. Low suction | Foam inside the jar | Fill the jar to 1/3 full of ordinary water |
| 8. No aspiration due to flow leakage of mucus | Filter blocked | Replace filter |
| 9. The Vacuum power on the patient side is either very low or absent | • Vacuum regulator set to minimum• Protection filter blocked or damaged• Connection tubes blocked, kinked or disconnected• Shut-off valve blocked or damaged• Pump motor damaged | • Turn the vacuum regulator clockwise and check the value of the vacuum on the gauge• Replace the filter• Replace or reconnect the tubes, check the jar connections• Empty the jar, or disconnect the tube from the jar and unblock the shut-off valve. The unit twill only work in the upright position• Contact the technical service |
| 10. Noisy | Technical internal problem | Contact the technical service |
| Faults 1 – 2 – 3 – 4 – 5 – 6 – 7 – 8 – 9 – 10 | None of the remedies has achieved the desired results | Contact the seller or GIMA After-sales Assistance Service |
If the overfill security system is activated, don’t proceed with the liquid aspiration. If the overfill security system doesn’t work there are two cases:
1st case – If the overfill security system doesn’t work the aspiration will be stopped by the bacteriological filter who avoids the liquid penetration inside the device.2nd case – If both the security system and the bacteriological filtrer do not work, there is the possibility that liquid has leaked inside the device, in this case return the device to GIMA technical service.
GIMA S.p.A. will provide upon request electric diagrams, components list, descriptions, setting instructions, and any other information that can help the technical assistance staff for productrepair.
BEFORE EVERY CHECKING OPERATION, IN CASE OF ANOMALIES OR BAD FUNCTIONING, PLEASE CONTACT GIMA TECHNICAL SERVICE. GIMA DOES NOT GIVE A GUARANTEE IF THE INSTRUMENT, AFTER THE TECHNICAL SERVICE CHECKING, APPEARS TO HAVE TAMPERED.
INSTRUCTION FOR USE
• The device must be checked before each use in order to identify any operating faults and/or damage due to transport and/or storage.· The work surface must be flat and stable in order that the control panel can be reached and so that the vacuum gauge, vase and antibacterial filter are clearly visible.· It is recommended not to hold the device in your hand and/or avoid prolonged contact with the body of the device.WARNING: For correct use, position the suction unit on a flat and stable surface, thus maximizing the available volume of the vase and improving the efficiency of the overflow device.
Operation with AC/DC power pack:
- Connect the short silicone tube with the antibacterial filter to the suction port. The other tube, connected to the filter on one end, must be connected to the spout on the vase lid with the float assembled inside (overflow device). The overflow device is triggered (the float closes off the internal lid fitting) when the maximum volume (90% of the effective vase volume) is reached, thus preventing the liquid from penetrating the inside of the machine. The device must be used on a flat, horizontal surface.
- Connect the long silicone tube to the free spout on the lid; the free end of the tube must be connected to the conical fitting for the probe coupling, to which the suction probe must then be connected.
- Connect the universal power pack to the device using the dedicated connector and insert the power cord plug into the socket. To start the treatment, press the switch into position I to turn the device on
- Set the desired vacuum level (Bar / kPa) through the vacuum regulator. Turn the knob in a clockwise direction to increase the vacuum level: these values can be read on the “vacuumgauge”.
- To suspend and/or end the treatment, press the switch again and pull the plug out of the socket
- To mitigate the formation of foam inside the vase, unscrew and remove the lid from the vase, and fill the latter with 1/3 water (to facilitate cleaning operations and speed up depressurization during operation), then screw the lid back onto the vase.
- Remove the accessories and proceed with cleaning operations.
- At the end of each use place the device back in the box, protected against dust
WARNING: The power cord plug is the element of separation from the electrical mains, even if the device is equipped with an on/off button. Once the device is in use, the power plug must remain accessible to allow another method of disconnection from the electrical mains.
Filter assembling![]() Make sure the filter is assembled with the arrows on the side of the patient.
Make sure the filter is assembled with the arrows on the side of the patient.
WARNING: The inside of the medical device must be regularly checked for the presence of liquidsor other visible contamination (secretions). In the presence of liquids or other visible contamination, immediately replace the medical device due to the risk of an insufficient vacuum flow rate.These products have been designed, tested and manufactured exclusively for single patient use andfor a period no longer than 24 hours.
Operation using cigarette lighter DC 12V
- Connect the device’s external plug 12V to the ligther plug with the cigarette lighter cable. Check the battery power status of the vehicle before the cigarette lighter cable. Press the switch to start suction
- Press the switch to the I position to turn it on.
WARNING: Only use the originally supplied or recommended replacement cigarette lighter cables(view the chapter “Important Safety Rules”)
Operation with Internal Battery
- Press the switch in position I to turn the device on (the external power pack doesn’t need to be connected)
- The fully charged battery life is about 60 minutes with continuous operation.
WARNING: Before using the device, check the battery power status. Before each use proceed with charging the battery. To maintain the device in good conditions, recharge the battery every 3 months (when not in use).
Recharging operations: to be able to charge the internal battery it is necessary to connect the universal switching adapter to the electric network for approx. 240 minutes with the main switch to position 0.
TAB. I – INDICATOR LIGHTS DURING OPERATIONS
When an external power supply is connected (regardless of the state of the battery charger) and when the device is working (after having turned it on), the LED stays in a FIXED GREEN position.
| LED Signal | Phase | Problem / Cause |
Solution |
| Flashing Green Led | During rechanrge | Battery recharge running | Wait |
| Steady Greed Led | During rechanrge | Recharging cycle complete | Remove power supply |
| Steady Red Led | During battery operation | Flat battery | Start recharging cycle
WARNING: During this signal, you will hear a long, continuous beep (duration of sound 0.8 sec/sound frequency: every 8.5 sec), which notifies the user regarding the battery discharge. |
| Flashing Red Led | Device automatically turns off when the battery is flat | Battery completely flat | When the device is restarted the LED will flash red: begin the battery recharge cycle immedi- ately |
| Steady Orange Led | During battery operation | Intermediate status | Guaranteed battery function / Recharge when the red LED signal comes on. |
NEVER USE THE DEVICE WITHOUT JAR AND / OR PROTECTION FILTERRISK OF ELECTROMAGNETIC INTERFERENCE AND POSSIBLE REMEDIESThis section contains information regarding the conformity of the compliance with the EN 60601-1-2 Standard(2015).The SUPER VEGA BATTERY PLUS SUCTION UNIT surgical aspirator is an electro-medical device that requires particular precautions regarding electromagnetic compatibility and which mustbe installed and commissioned according to the electromagnetic compatibility information supplied. Portable and mobile radio communication devices (mobile phones, transceivers, etc.) may interfere with the medical device and should not be used in close proximity with, adjacent to or on top of the medical device. If such use is necessary and unavoidable, special precautions should be taken so that the electro-medical device functions properly in its
intended operating configuration (for example, constantly and visually checking for the absence of anomalies or malfunctions). The use of accessories, transducers, and cables differing from those specified, with the exception of transducers and cables sold by the appliance and system manufacturer as spare parts, can lead to an increase in emissions or in a decrease of the immunity of the device or system. The following tables supply information regarding the EMC (Electromagnetic Compatibility) characteristics of the electro-medical device.
| Guidance and manufacturer’s declaration – electromagnetic Emissions | ||
| The SUPER VEGA BATTERY PLUS SUCTION UNIT is intended for use in the electromagnetic environment specified below. The customers or the user of the SUPER VEGA BATTERY PLUS SUCTION UNIT should make sure that it’s used in such an environment. | ||
|
Emissions test |
Compliance |
Electromagnetic environment – guidance |
| Irradiated / Conducted emissions CISPR11 | Group 1 |
The SUPER VEGA BATTERY PLUS SUCTION UNIT only used RF energy only for its internal functioning. Therefore, its RF emissions are very low and are not cause interference in the proximity of any Electronic appliances. |
| Irradiated / Conducted emissions CISPR11 | Class [B] | The SUPER VEGA BATTERY PLUS SUCTION UNIT can be used in all environments, including domestic and those connected directly to the public mains distribution that supplies power to environments used for domestic scopes. |
| Harmonic emissions EN 61000-3-2 | Class [A] | |
| Voltage fluctuations / flicker emissions EN 61000-3-3 | Complies |
| Guidance and manufacturer’s declaration – Immunity Emissions | |||
| The SUPER VEGA BATTERY PLUS SUCTION UNIT is intended for use in the electromagnetic environment specified below. The customers or the user of the SUPER VEGA BATTERY PLUS SUCTION UNIT should make sure that it’s used in such an environment. | |||
| Immunity Test | Level indicated by the EN 60601-1-2 | Compliance Level | Electromagnetic environment – guidance |
| Electrostatic discharge (ESD) EN 61000-4-2 | +/-8kV on contact+/-15kV in air | The device doesn’t change its state | Floors should be wood, concrete or ceramic tile. If floors are covered with synthetic material, the relative humidity should be at least 30%. |
| Electrical fast transient/burst EN 61000-4-4 | ± 2kV power supply lines ± 1kV for input/output lines | The device doesn’t change its state | Mains power quality should be that of a typical commercial environment or hospital. |
| Surge 61000-4-5 | ± 1kV differential mode +/-2 kV ordinary mode | The device doesn’t change its state | Mains power quality should be that of a typical commercial environment or hospital. |
| Loss of voltage, brief voltage interruptions and variations61000-4-11 | <5% UT (>95% dip UT ) for 0,5 cycle 40% UT (60% dip UT ) for 5 cycle70% UT (30% dip UT ) for 25 cycle <5% UT (>95% dip UT) for 5 sec | – – | Mains power quality should be that of a typical commercial environment or hospital If the user of the SUPER VEGA BATTERY PLUS SUCTION UNIT request that the appliance operates continuously, the use of a continuity unit is recommended. |
| A magnetic field with network frequency (50/60 HZ)61000-4-8 | 30A/m | The device doesn’t change its state | The power frequency magnetic field should be measured in the intended installation location to make sure that it’s sufficiently low. |
|
Note UT is the value of the power supply voltage |
| Guidance and manufacturer’s declaration – Immunity Emissions | |||
| The SUPER VEGA BATTERY PLUS SUCTION UNIT is intended for use in the electromagnetic environment specified below.
The customers or the user of the SUPER VEGA BATTERY PLUS SUCTION UNIT should make sure that it’s used in such an environment. |
|||
|
Immunity Test |
Level indicated by the EN 60601-1-2 | Compliance Level |
Electromagnetic environment – guidance |
| Conducted Immunity61000-4-6Radiated Immunity 61000-4-3 | 3Vrms 150kHz to 80Mhz (for non-life-supporting devices)10V/m 80MHz to 2.7GHz (for non-life-supporting devices) | V1 = 3 V rmsE1 = 10 V / m | The portable and mobile RF communication devices, including cables, must not be used closer to the SU- PER VEGA BATTERY PLUS SUCTION UNIT device, than the separation distance calculated by the equation applicable to the transmitter frequency.Recommended separation distanced= 3,5 √P V 1d= 12 √P from 80 MHz to 800MHzE 1d= 23 √P from 800 MHz to 2.7 GHzE 1Where P is the maximum nominal output voltage of the transmitter in Watt (W) depending on the manufacturer of the transmitter and the recommended separation distance in meters (m). The intensity of the field from the fixed RF transmitters, as determined by an elec- the tro-magnetic study of the sitea), could be lower than the level of conformity of each frequency interval b).It is possible to check for interference in proximity to devices identified by the following symbol: |
|
Note 1: At 80 MHz and 800 MHz the interval with the highest frequency is applied Note 2: These guidelines may not be applicable in all situations. The electromagnetic propagation is influenced by absorption and by reflection from buildings, objects and people. |
|||
|
a) The field intensity for fixed transmitters such as the base stations for radiotelephones (mobile and cordless) and terres- trial mobile radio, amateur radio devices, radio AM and FM transmitters and TV transmitters can not be theoretically and accurately foreseen. To establish an electro-magnetic environment generated by fixed RF transmitters, an electro-magnetic study of the site should be considered. If the field intensity measured in the place where the device will be used surpass- es the above-mentioned applicable level of conformity, the normal functioning of the device should be monitored. If abnormal performance arises, additional measures such as changing the device’s direction or positioning may be necessary.b) The field intensity on an interval frequency of 150 kHz to 80 MHz should be less than 10 V/m. |
|
The recommended separation distance between portable and mobile radio-communication devices and the monitor |
|||
|
The SUPER VEGA BATTERY PLUS SUCTION UNIT surgical aspirator is intended to operate in an electro-magnetic environment where RF irradiated interferences are under control. The client or operator of the SUPER VEGA BATTERY PLUS SUCTION UNIT device can help prevent electro-magnetic interference by keeping a minimum distance between the portable and mobile RF communication devices (transmitters) and the SUPER VEGA BATTERY PLUS SUCTION UNIT device, as recommended below, in relation to the radio-communication maximum output power. |
|||
| The maximum nominal output power of the Transmitter W |
Separation distance from the frequency transmitter (m) |
||
| 150KHz to 80MHz
d= 3,5 √P V 1 |
80MHz to 800MHz
d= 12 √P E 1 |
800MHz to 2,7GHz
d= 23 √P E 1 |
|
| 0,01 | 0,12 | 0,12 | 0,23 |
| 0,1 | 0,38 | 0,38 | 0,73 |
| 1 | 1,2 | 1,2 | 2,3 |
| 10 | 3,8 | 3,8 | 7,3 |
| 100 | 12 | 12 | 23 |
|
For transmitters with a maximum nominal output power not shown above, the recommended separation distance in meters (m) can be calculated using the equation applicable to the transmitter frequency, where P is the maximum nominal output power of the transmitter in Watt (W) depending on the transmitter’s manufacturer.Note 1: At 80 MHz and 800 MHz the interval with the highest frequency is appliedNote 2: These guidelines may not be applicable in all situations. The electromagnetic propagation is influenced by the absorption and by the reflection from buildings, objects, and people |
SYMBOLS
![]()
![]() Disposal: The product must not be disposed of along with other domestic waste. The users must dispose of this equipment by bringing it to a specific recycling point for electric and electronic equipment.
Disposal: The product must not be disposed of along with other domestic waste. The users must dispose of this equipment by bringing it to a specific recycling point for electric and electronic equipment.
DISPOSAL OF WASTE BATTERIES – (Directive 2006/66/EC) This symbol on the battery or on the packaging indicates that the battery provided with this product shall not be treated as ousehold waste. By ensuring these batteries are disposed of correctly, you will help prevent potentially negative consequences for the environment and human health which could otherwise be caused by inappropriate waste handling of the battery. The recycling of the materials will help to conserve natural resources. At the end of their life hand the batteries over to the applicable collection points for the recycling of waste batteries. For more detailed information about recycling of this product or battery, please contact your local Civic Office, your household waste disposal service or the shop where you purchased the product.
GIMA WARRANTY TERMSThe Gima 12-month standard B2B warranty applies.
References
[xyz-ips snippet=”download-snippet”]

