User Manual

Viatom Pulse Oximeter FS10C/FS20C
Shenzhen Viatom Technology Co., Ltd.Add: 4E, Building 3, Tingwei Industrial Park, HonglangNorth 2nd Road, Baoan, 518100 Shenzhen,Guangdong, P.R.ChinaWebsite: www.viatomtech.comEmail: Tel: 86-755-86638929 Fax: 86-755-22649904
 Hunan Accurate Bio-Medical Technology Co., Ltd Add: 6th,Floor,Biyang Industrial Zone, Lijiacun Road,Xueshi Street of Yuelu District,410208 Changsha,Hunan Province,PEOPLE’S REPUBLIC OF CHINA Tel/ Fax: +86 -731-84118539
Hunan Accurate Bio-Medical Technology Co., Ltd Add: 6th,Floor,Biyang Industrial Zone, Lijiacun Road,Xueshi Street of Yuelu District,410208 Changsha,Hunan Province,PEOPLE’S REPUBLIC OF CHINA Tel/ Fax: +86 -731-84118539
EC REPRESENTATIVE Shanghai International Holding Corp. GmbH (Europe)Add: Eiffestrasse 80, 20537 Hamburg,GermanyTel: +49-40-2513175 Fax: +49-40-255726
Instructions to User
Dear users, thank you very much for purchasing the Pulse Oximeter. This manual contains the instructions necessary to operate the product safely and in accordance with its function and intended use. Observance of this manual is a prerequisite for proper product performance and correct operation and ensures patient and operator safety. Read this manual carefully before using the fingertip pulse oximeter. This product is reusable medical device. The using life is 2 years. The device is prescription use device.
1. Safety
Contraindications Do NOT use oximeter in a magnetic resonance (MR) environment.
Warnings Keep the oximeter away from young children. Small parts such as the battery door, battery and lanyard may trigger choking hazards.
Cautions
- Do not use oximeter in the present of flammable anesthetics.
- The oximeter needs to be used according to information provided in the user manual.
- The equipment is NOT intended for neonate and infant.
- Do not use a damaged oximeter which may affect measurement performance.
- Do not place the oximeter on the same hand/arm when suing a blood pressure cuff or monitor.
- Do not use the oximeter for more than 30 minutes without relocating the device to another finger.
- Do not place the oximeter on edema or fragile tissues.
- Do not use the oximeter as the only basis for making medical decisions, it is intended only to be used as addtional information that you can give to your licensed health care professional.
- Do not use the oximeter in high frequency environment such as electrosurgical equipment.
- Do not place the oximeter in liquid.
- Follow local disposal and recycling laws for the oximeter and its components, including the battery.
- Do not stare the light (the infrared is invisible) which emitted from the oximeter is harmful to the eyes.
- The oximeter is designed to measure the percentage of arterial oxygen saturation of functional hemoglobin. Any of the following conditions may reduce the performance of the oximeter.
- Flicking or very bright light
- Moisture in the oximeter
- The individual weight less than 10kg
- Week pulse quality(low perfusion)
- Venous pulsations
- Low hemoglobin
- Cardiogreen and other intravascular dyes
- Carboxyhemoglobin
- Methemoglobin
- Dysfunctional hemoglobin
- Artificial nails or fingernail polish
2. The Basics
The pulse oxygen saturation is the percentage of HbO2 in the total Hb in the blood, so-called the O2 concentration in the blood. It is an important bio-parameter for respiration. A number of diseases relating to respiratory system may cause the decrease of SpO2 in the blood, furthermore, some other causes such as the malfunction of human body’s self-adjustment, damages during surgery, and the injuries caused by some medical checkup would also lead to the difficulty of oxygen supply in human body. And the corresponding symptoms would appear as a consequence, such as vertigo, impotence, vomit etc. serious symptoms might bring danger to human’s life. Therefore, prompt information of patients Spo2 is of greats help for the doctor to discover the potential danger, and is of great importance in the clinical of medical field.
Principle
Principle of the oximeter is as follows: An experience formula of data process is established taking use of Lambert Beer Law according to Spectrum Absorption Characteristic of Reductive hemoglobin (Hb)and oxyhemoglobin(HbO2)in glow & near-infrared zones. Operation Principle of the device is: Photoelectric Oxyhemoglobin Inspection Technology is adopted in accordance with Capacity Pulse Scanning Recording Technology, so that two beams of different wavelength of lights can be focused onto human nail tip through perspective clamp finger-type sensor. Then measured signal can be obtained by a photosensitive element, information acquired through which will be shown on screen through treatment in electronic circuits and microprocessor.
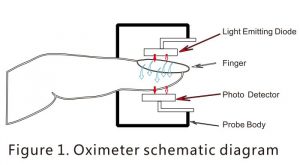
Introduction
Intended Use The Pulse Oximeter is portable, convenient, non-invasive devices, used for monitor arterial hemoglobin oxygen saturation (SPO2) and pulse rate. The personal application are adult patients (weight: > 30kg) and pediatric patients (weight: 20-30kg). We recommend index finger, middle finger and ring finger are suitable position for monitor. It’s intended for spot-checking or attended-care monitoring in Home Health Care and Medical Facility.
WARNING:
- This pulse oximeter is intended for use only by clinical professionals or under their guidance. It must only be used by persons who have received adequate training in its use. Anyone unauthorized or untrained must not perform any operation on it.
CAUTIONS:
- This pulse oximeter is intended for use in hospital,clinical institution, healthcare community.
- The pulse oximeter is NOT designed for newborn and infant. For adults and children, it recommended that the finger thickness should between 8-25.4mm.
NOTES
- The probe is the hole in the middle of the equipment to which the finger insert.
- The probe is the Applied Part of the equipment.
Features
- The pulse oximeter is small in volumes, light in weight and easy to carry.
- Low in power consumption, 600 spot-checks on two AAA batteries.
- One button and easy to operate.
- There are three modes: power off, sleep and measure.
- Automatically turning into sleep mode within 8 seconds after there is no signal.
NOTES
- Press the operating button to activate oximeter (measure mode) from sleep mode.
- Power off after removing the batteries.
Front View

Functions

Symbols

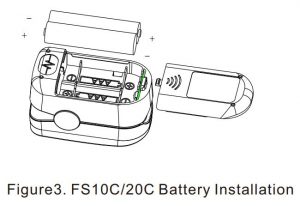
3. Battery Installation
A. Put the two AAA batteries into battery compartment in correct polarities. B. Push the battery cover horizontally along the arrow shown as right.
WARNINGS:
- Battery polarities should be correctly installed, otherwise, damage may be caused to the equipment.
- Please remove the batteries if the oximeter will be stored for more than 30 days.
- Please remove the batteries if you want turn off the oximeter otherwise it is always in power state.
- Battery may leak or explode if used or disposed off improperly
4. Operating Guide
Application Method
- Remove the battery cover, and insert the two AAA batteries following polarity markings indicated inside of the battery compartment, then reposition the cover;
- Hold the oximeter with the display facing toward you, slide your finger into the opening probe of the device, as shown below (Figure 4), until the fingertip touches the built-in stop guide. For best results, make sure the finger is centered within the finger guide.
- Press the button to activate the oximeter from sleep mode, and then measurement interface will appear in 3 seconds.
- The measurement result will be read directly from the screen within 10 seconds;
- The oximeter will turn into sleep mode automatically within 8 seconds after the finger left the probe.

Attention for Operation
- Ring finger, middle finger and index finger are recommended as suitable monitor position.
- Excessive or rapid movement may affect measurement.
- Improper sensor placement may affect the measurement accuracy.
- The oximeter can be reused after cleaning and distinfection.
- The measurement comes to the best when the oximeter and the heart are at the same level.
- (Only for FS20C) The plethysmogram can be used as signal quality indicator. The displayed parameters might be unreliable with the disorderly plethysmogram.
- (Only for FS10C) The bar graph can be used as signal quality indicator. The displayed parameters might be unreliable with un-periodic bar change.
- The displayed parameters will show invalid indicator as ‘—‘if signal quality is very low.
- The displayed parameters will show invalid indicator as ‘—‘if oximeter fault ouccur.
- The maximum continuous test time does not exceed 5 minutes.
Specifications


6. Technical Description
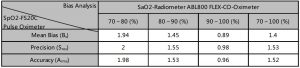
The below table shows statistic conclusion of an invasive controlled desaturation study which guided by “ISO80601-261-2011, Annex EE, Guideline for evaluating and documenting SpO2 Accuracy in human subjects’.
The statistic result displayed the accuracy distribution between the range of 70%-100%, which may helpful to user. Bias Analysis
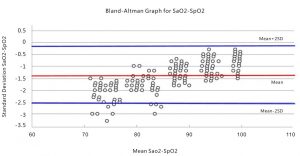
The below is the Bland-Altman graphical plot of samples from invasive controlled desaturation study.
7. Maintenance, Cleaning, Disinfection
Maintenance
The equipment’s design life expectancy is about 2 years, keep your equipment and accessories free of dust and dirt, and follow these rules:
- Please clean the equipment before use according to chapter 6.2;Remove the batteries inside the battery cassette if the equipment will not be operated for a long time;
- Replace the batteries in time when the battery voltage indicate lamps were empty;
- It is recommended that the equipment should be kept in a dry environment with no corrosive gases and good ventilation anytime. The moisture and high-light environments will affect its lifetime and even might damage the equipment.
- It is best to preserve the product in a place where the temperature is between -20 to 60°C and the relative humidity is less than 95%.
- The packed equipment can be transported by ordinary conveyance. The equipment not be transported mixed with toxic, harmful, corrosive materials.
WARNING
- No modification of this equipment is allowed.
Disposal Dispose of the pulse oximeter in accordance with local environment and waste disposal laws and regulations.
8. Cleaning / Disinfection
CAUTIONS
- Never immerse or soak the oximeter.
- We recommend that the oximeter be cleaned and disinfected after use every time or determined by your hospital’s policy, to avoid long term damage to the oximeter.
- Never use cleaning agents/disinfectants other than the recommended.
- The sensor component is not cleaned and disinfected during testing.
Cleaning
The recommended cleaning agents include: water
- Shut down the pulse oximeter and remove the battery.
- Clean the oximeter with cotton or soft cloth moistened with water.
- After cleaning, wipe off the water with a soft cloth.
- Allow the oximeter to air dry.
Disinfection
The recommended disinfectants include: ethanol 70%, isoppropanol 70%.
- Shut down the pulse oximeter and remove the battery.
- Clean the oximeter as instructed above.
- Disinfect the oximeter with cotton or soft cloth moistened with one of the recommended disinfectants.
- After disinfection, be sure to wipe off the disinfectant left on the oximeter with a soft cloth moistened with water.
- Allow the oximeter to air dry.
9. Accessories
- One lanyard.
- Two AAA batteries.
- One user manual
- One certificate card
Note:
- For particular configuration of accessories please refer to the product package list.
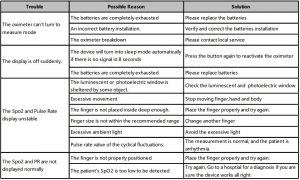
10. Troubleshooting
WARNINGS
- Necessary maintenance must be performed by qualified service personal ONLY.
- Users are NOT permitted to maintain the equipment by themselves.
- There are NO replaceable components in the equipment.
Appendix A
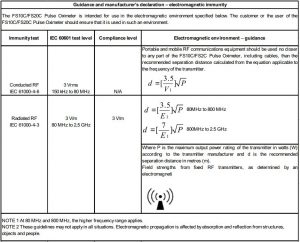
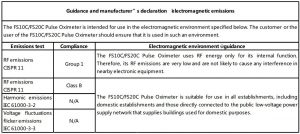
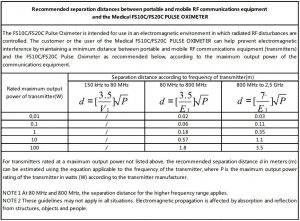
The equipment complies with the requirement of standard EN 60601-1-2:2007 “Electromagnetic Compatibility – Medical Electrical Equipment”.

Viatom Pulse Oximeter FS10C/FS20C User Manual – Viatom Pulse Oximeter FS10C/FS20C User Manual –
Questions about your Manual? Post in the comments!
[xyz-ips snippet=”download-snippet”]

